Prebiotics are big business, their production and commercialisation for the functional food industry driven by consumer engagement around the impact of nutrition on health and wellbeing.
In 2021 the prebiotics market was valued at $US6.05 billion globally and is expected to rise at a compound annual growth rate (CAGR) of 14.9% from 2022 to 2030.1
As more becomes known about the positive links between the gut microbiome and overall health, research continues to show that prebiotics can play a role in promoting healthy body functions, including digestion and metabolic health. Yet with numerous prebiotics products on the market, consumers can struggle to make informed choices about those which will best suit their needs.
Galacto-oligosaccharides (GOS)
One group of prebiotics is galacto-oligosaccharide (GOS), a type of oligosaccharide. Derived from dairy products, some root vegetables and beans, GOS is resistant to the acidic pH of the stomach and is known to promote the growth and activity of good bacteria in the gut, thereby benefitting the host’s health.
GOS has also been shown to significantly stimulate bifidobacteria and lactobacilli – the lactic acid bacteria (LAB) that are important in the gut microbiota – and bifidobacteria in infants have shown high incorporation with GOS. The findings of a 2020 study indicated that oral administration of GOS promoted the proliferation of LAB in the intestinal flora, as well as short chain fatty acid (SCFA) production, the effect of which increased moisture content of faeces and alleviated constipation in subjects.2
GOS prebiotics have a range of properties that make them suitable as a component in food manufacturing, in applications including syrups, gummies, capsules and dissolvable powders. They have little to no effect on the taste of food and, unlike other probiotics and prebiotics that are pH-sensitive, GOS is not destroyed during the manufacturing process and remains stable until it reaches the digestive tract, which is an important factor in functional foods.
In addition, GOS has excellent heat stability and is economically and conveniently beneficial for manufacturers due to its stability food processing, storage, and transportation. Neo Cremar’s acid resistance test confirmed that GOS was not destroyed at pH 2, 3, and 4, and it is a stable and convenient option for food manufacturers.
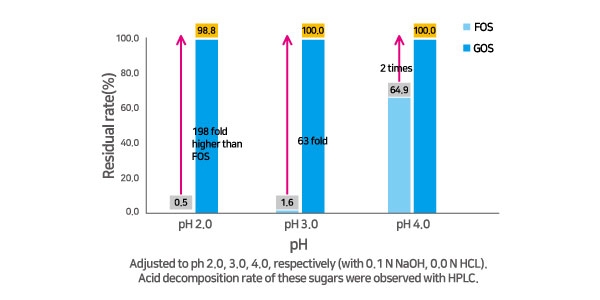
GOS also have a high solubility rate which offers potential across a range of applications and formulations, and can promote synergy with other prebiotics and probiotics during the manufacturing process in functional foods.
Functional fortification for skin health
Many companies and research institutes are conducting various studies on GOS to date. Neo Cremar has also conducted research on promoting gut health as well as preventing and improving skin damage caused by UV radiation.
NeoGOS has been shown to improve skin health issues, such as erythema and pigmentation, by having a positive effect on the intestinal environment, which in turn increases antioxidant enzymes and leads to significant retention of collagen levels (by inhibiting collagen-degrading enzymes). Clinical studies have shown that supplementation of NeoGOS increased skin moisture content and reduced the number and depth of wrinkles by 12.2% within four weeks.
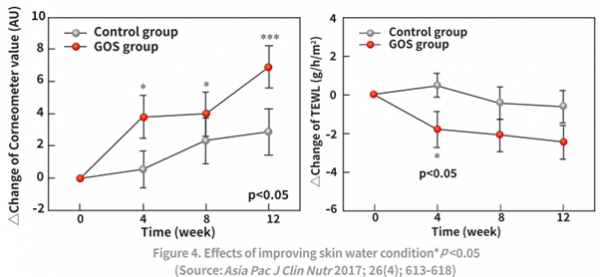
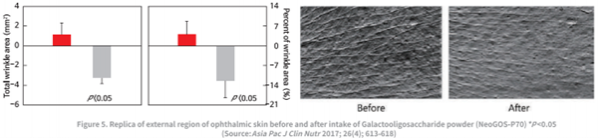
In conclusion, these studies show that consuming 2g of NeoGOS per day can increase skin hydration and reduce the total area and distribution of wrinkles, thus improving skin health. Ultimately, this can help reduce skin damage that may occur from daily exposure to UV rays.3
As a result of these studies, NeoGOS has been approved by the South Korean Ministry of Food and Drug Safety as an individually Recognized Functional Ingredient (IRFI), allowing finished products using this ingredient to make health claims such as ‘helping to improve skin health by protecting against skin damage caused by UV radiation’. Additionally, NeoGOS has also been registered under Japan’s Functional Food Labelling System.
NeoGOS – future innovations
Established in 1999, Neo Cremar began producing Korean-made GOS in 2014 after developing it through research. As a company that pursues health for people, not just a simple food ingredient supplier, Neo Cremar has constantly improved the quality and functionality of GOS through various studies.
Despite being a relatively latecomer compared to overseas manufacturers in GOS production and development, Neo Cremar had already completed development and conducted clinical trials of HMO-enhanced GOS named Mother’s Oligo in the mid-2010s when HMO-enhanced GOS was commercialized. Furthermore, in 2017, it was the first to launch organic GOS, which still maintains the highest quality since its launch.
While GOS can meet various formulation requirements of food and beverage producers, NeoGOS has become a product that can meet even broader formulation requirements and product concepts, thanks to various added functionalities through repeated studies. NeoGOS has been applied to various types of products and formulations produced by many food and beverage manufacturers worldwide and continues to release new products.
Recently, Neo Cremar conducted studies in the field of research, focusing on the improvement of inflammatory bowel disease through the intake of galacto-oligosaccharides and the efficacy of allergic dermatitis symptoms, obtaining significant results. Keeping up with the latest market trends, Neo Cremar has completed the development of high-purity GOS with a higher content than the current product and plans to launch it soon.)
The Neo Cremar team will be attending Vitafoods Europe in Geneva in May where they will showcase the health benefits of their prebiotics portfolio within the functional food and infant formula markets, and demonstrate their ease of use.
To find out more, visit Vitafoods Europe 9-11 May 2023, or get in touch via Neo Cremar.
References
1. Prebiotics Market Size, Share & Trends Analysis Report By Ingredients (FOS, Inulin, GOS, MOS), By Application (Food & Beverages, Dietary Supplements, Animal Feed), By Region, And Segment Forecasts, 2022 – 2030. Grand View Research.
2. Guk Kim M.; Kyungae Jo K;, Boo Chang Y.; et al. Changes in the Gut Microbiome after Galacto-Oligosaccharide Administration in Loperamide- Induced Constipation. Journal of Personalized Medicine (2020, October). Department of Integrated Biomedical and Life Science, Graduate School, Korea University, Seoul 02841, Korea.
3. Hong, Y. H.; Chang, U. J.; Kim, Y. S.; et al. Dietary galacto-oligosaccharides improve skin health: A randomized double blind clinical trial. Asia Pacific Journal of Clinical Nutrition, 26(4), 613–618 (2017).




